
Posted by2 years ago
Archived
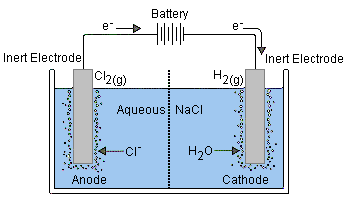
Brother mfc-l3770cdw driver windows xp. During electrolysis of aqueous zinc chloride what will be the products that can be seen at the anode and cathode? In MOLTEN zinc chloride, At the cathode: Zn2+ + 2e- - Zn(s).
How do the products compare at each electrode when aqueous 1 mol dm-3 magnesium bromide and molten magnesium bromide are electrolysed?
Mg2+ + 2e- <=> Mg(s) [V = -2.37]
0.5Br2 (l) + e- <=> Br- (aq) [V = +1.07]
0.5O2(g) + 2H+ (aq) + 2e- <=> H2O (l) [V = +1.23]

Negative Electrode: Same or Different Positive Electrode: Same or Different
I thought Mg2+ would be attracted to the negative electrode in both the molten and aqueous state and that the Br ion would be attracted to the positive electrode in the molten state but the OH would overtake the Br ion's attraction in the aqueous state, giving Same for the negative electrode and Different for the positive electrode.
Dante virtual sound card bloque dans mac. However, the given answer simply states Same for the positive electrode and Different for the negative electrode. Why so?
1 comment

